José Vilar and Friends Case 49 (Solution+Teaching Tips!)
This case was sent to me by Dr. Ana Villanueva, a young Spanish radiologist actually training at Sunnybrook Health Sciences Centre (Toronto). 
Ana is a good representation of the new generation of radiologists. Her father, Dr. Alberto Villanueva has contributed to this blog (Case 31) and probably is partly responsible for Ana´s interest in thoracic Radiology.
This is a case from Sunnybrook Health Sciences Centre in Toronto, Canada
Dr Mohamed Saber and Assoc Prof Frank Gaillard et al.
Case 49:
16 year-old female
Reason for the study: history: cough, shortness of breath, right sided chest pain, fever x 3 days
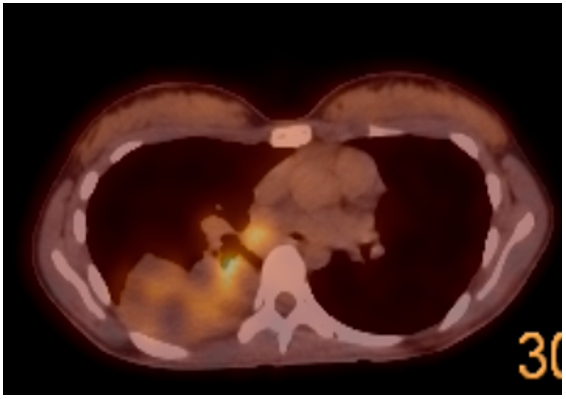
Update: I’ll include CT and a PET/CT image.
Can you now think of possible diagnosis?
Click here for the answer
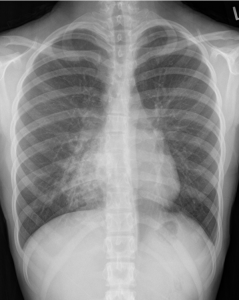
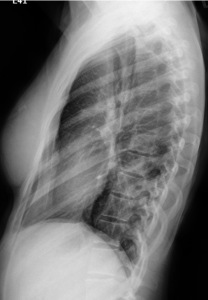
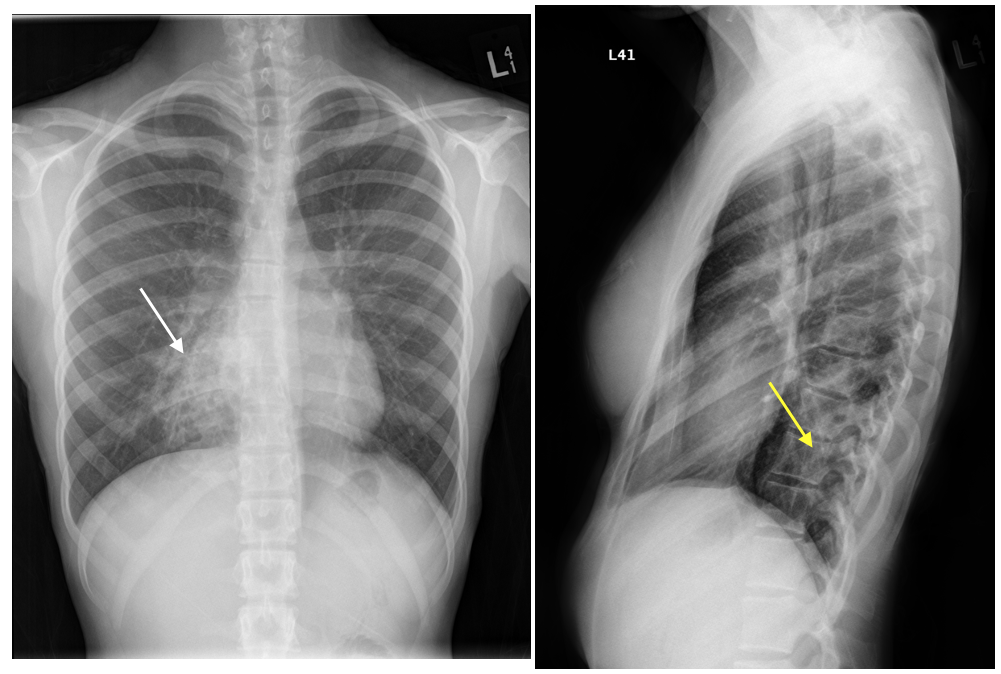
Findings: In the PA projection there is an opacity in the right lower lobe. The pathology does not blur the right heart border (white arrow) indicating that is posterior as can also be confirmed in the lateral projection. (Yellow Arrow).
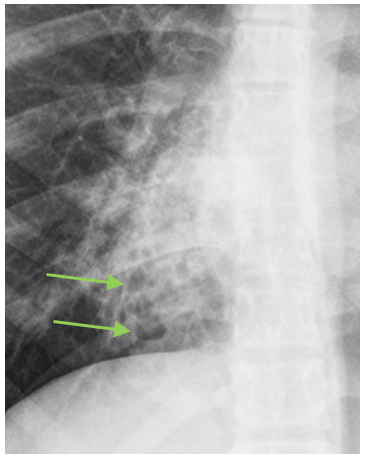
Additionally, there are some “cystic areas” within the opacity that were mentioned by some of you (Elena and Sofija).
( Green arrows)
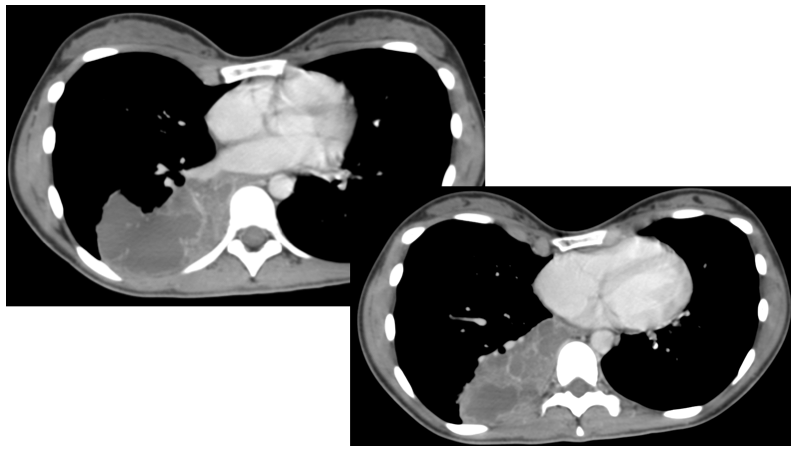
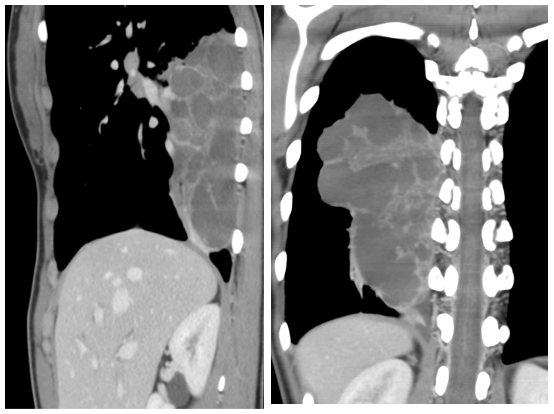
In the CT there is a mass like lesion occupying part of the right lower lobe. It contains cystic areas of water density. No systemic arterial supply was detected (Not shown).
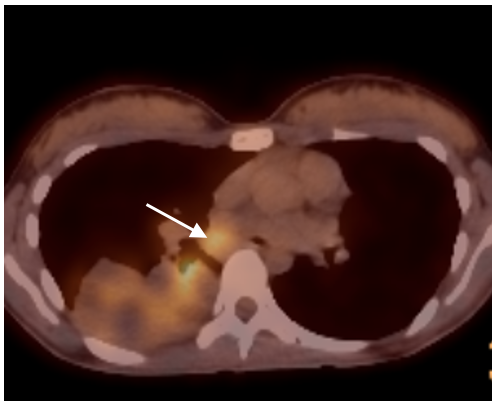
The PET/CT revealed metabolic activity within the lesion and in the mediastinum (white arrow).
Right lower lobe lobectomy,
THE HISTOLOGICAL APPEARANCE IS CONSISTENT WITH CONGENITAL PULMONARY AIRWAY MALFORMATION (CPAM) SECONDARILY INFLAMMED
Congenital pulmonary airway malformation (Type I)
( Well done Marcos Mestas!).
Congenital pulmonary airway malformations (CPAM) are multicystic masses of segmental lung tissue with abnormal bronchial proliferation. CPAMs are considered part of the spectrum of bronchopulmonary foregut malformations.
Terminology
Until recently they were described as congenital cystic adenomatoid malformations (CCAM).
Epidemiology
They account for ~25% of congenital lung lesions. The estimated incidence is approximately 1:1500-4000 live births and there is a male predominance.
Clinical presentation
The diagnosis is usually either made on antenatal ultrasound, or in the neonatal period on the investigation of progressive respiratory distress 3,4. If large, they may cause pulmonary hypoplasia, with resultant poor prognosis.
In cases where the abnormality is small, the diagnosis may not be made for many years or even until adulthood. When it does become apparent, it is usually as a result of recurrent chest infection 3,4.
Pathology
The condition results from failure of normal bronchoalveolar development with a hamartomatous proliferation of terminal respiratory units in a gland-like pattern (adenomatoid) without proper alveolar formation.
Histologically, they are characterized by adenomatoid proliferation of bronchiole-like structures and macro- or microcysts lined by columnar or cuboidal epithelium and absence of cartilage and bronchial glands.
These lesions have intracystic communications and, unlike bronchogenic cysts, can also have a connection to the tracheobronchial tree.
Subtypes
Five subtypes are currently classified, mainly according to cyst size:
- type I
-
- most common: 70% of cases 3
- large cysts
- one or more dominant cysts: 2-10 cm in size
- may be surrounded by smaller cysts
- type II
-
- 15-20% of cases 3
- cysts are <2 cm in diameter
- associated with other abnormalities
- type III
-
- ~10% of cases
- microcysts: <5 mm in diameter
- typically involves an entire lobe
- has a poorer prognosis
- type IV
-
- unlined cyst
- typically affects a single lobe
- indistinguishable from type I on imaging 11
- type 0
-
- very rare, lethal postnatally
- acinar dysgenesis or dysplasia 11
- represents global arrest of lung development 12
Location
Lesions are usually unilateral and involve a single lobe. Although there is no well-documented lobar predilection, they appear less frequently in the middle lobe .
Associations
- hybrid lesion: i.e. CPAM and pulmonary sequestration
- renal agenesis7
- polyhydramnios
- hydrops fetalis
- lung malignancy
-
- ~10% of pediatric lung cancers have a history of CPAM 13
- bronchoalveolar carcinoma associated with type 1 CPAM 14
- pleuropulmonary blastoma associated with type 4 CPAM 14
Radiographic features
The appearance of CPAMs will vary depending on the type.
Antenatal ultrasound
CPAM appears as an isolated cystic or solid intrathoracic mass. A solid thoracic mass is usually indicative of a type III CPAM and is typically hyperechoic. There can be a mass effect where the heart may appear displaced to the opposite side. Alternatively, the lesion may remain stable in size, or even regress 5.
Hydrops fetalis and polyhydramnios may develop and may be detected on ultrasound as ancillary sonographic features 3.
Plain radiograph
Chest radiographs in type I and II CPAMs may demonstrate a multicystic lesion. Large lesions may cause a mass effect with resultant mediastinal shift, depression, and even inversion of the diaphragm. In the early neonatal period, the cysts may be completely or partially fluid-filled, in which case the lesion may appear solid or with air-fluid levels. Lesions may change in size on interval imaging (expand from collateral ventilation via pores of Kohn). Type III lesions appear solid.
CT
CT has a number of roles in the management of CPAMs. First, it more accurately delineates the location and extent of the lesion. Secondly, and most important in surgical candidates, CT angiography is able to identify systemic arterial supply if present.
Appearance reflects the underlying type, and a type III lesion can appear as a consolidation.
Treatment and prognosis
There can be a wide spectrum of prognosis.
Surgery (elective lobectomy) is the treatment of choice in symptomatic patients, both in those presenting early with respiratory compromise and in those presenting later with recurrent infections 3. Type I lesions have the best prognosis.
In the setting of a small stable asymptomatic lesion, surgical excision is more controversial. Advocates for excision quote the reported risk of developing malignancies within the lesion (see above). An alternative approach is to watch and wait. There are reports of spontaneous regression, particularly in those serially followed up on antenatal ultrasound 7,10.
Complications
Potential postnatal complications include:
- recurrent pneumothorax
- hemopneumothorax
- pyopneumothorax
- possible incidence of certain malignancies, which include 3:
Potential in utero complications include:
- hydrops fetalismay rarely develop when there is severe compression of the fetal heart or great vessels
- compression of the normal fetal lung can also rarely cause pulmonary hypoplasia
Differential diagnosis
-
- Unilocular
- does not usually communicate with the bronchial tree, and are therefore typically not air-filled
-
- systemic arterial supply
- hybrid lesions may present with both CPAM and sequestration features (see above)
-
- bowel loops within a hemithorax
- congenital lobar emphysema(congenital lobar overinflation)
-
- hyperlucent and hyperinflated lung segment
- no cystic or solid components
- localized congenital cystic bronchiectasis
For type I lesions on CT also consider:
- cicatrisation collapsewith scarring and traction bronchiectasis
References
Preethi Chandrasekaran et al., Case Rep, 2021
Pierre Goussard et al., Case Rep, 2020
Rebecca Finnegan et al., Archives of Disease in Childhood, 2019
Amel Ben Hamed et al., Archives of Disease in Childhood, 2019
Rugo HS, et al., Oncologist, 2021
Bethany Taylor et al., Thorax, 2020
Tips: Dr Ana Villanueva thinks that two points have to be stressed in this case:
- The age as a factor that should always make you think of congenital pathology.( Especially if you are not a pediatric radiologist).
- The presence of bubbles or cysts in a pulmonary lesion also can narrow the differential diagnosis
I fully agree with Dr. Ana Villanueva and I think her and Drs Saber and Gaillard ( Sunnybrook Health Sciences Centre) for this wonderful case and complete discussion.




Right middle lobe pneumonia
x ray chest pa view and lateral views shows the following features;
slight scoliosis upper dorsal region
slightly vertical directed anterior ribs and slightly horizontal post end of ribs, depressed lower end of sternum with reduced ap diameter of chest in lateral view
indistinct rt cardiac border, cardiac silhoutte pushed to left side.
hyperventilated lung fields with wedge shaped opacity wit muliple small cystic space at right lower zone and cardiophrnic angle, spine sign in lateral view suggesting lower lobe pathology.
medial breast margin sign on left side
additional rib -lumbar rib on left side
features of pectus excavatum, with mild scoliosis,lower lobe pneumonia with bronchiectatic changes, with rib anomaly
Anomalie radiologique au níveau de D7 suggestive de spondilodiscite?
Neumonía del lóbulo medio
Mal de pott
Infected pulmonary sequestration
Right inferior lobe pneumonia
Pectus excavatum mimics middle lobe pneumonia.Right inferior lobe pneumonia
Wow, many people have dared participate!.A couple of coments regarding your ideas:1) Yes there is a pectus excavatum that could mimic right lung pathology or even sometimes cardiac pathology, but this is not the important finding in case 49. 2) And no, the pathology is not in the middle lobe, (just look at the lateral), its posterior, Now a bit of help: can you identify a specific pattern in the right lower lobe pathology?
Mass (extrapulmonary?)lesion.
Air fluid levels?
There are some gas inclusions in the projection of right cardiophrenic angle? Maybe in the thoracic wall?
Round pneumonia RLL
Necrotizing pneumonia of the right middle lobe
On the CT we confirmed what was evident on the lateral chest xray, the spine sign with some cystic areas in the right lower lobe.
GIven the image characteristics (multicystic opacity) and the age of the patient my first diagnosis would be a superinfected CPAM (type I).
There isn’t any arterial vessel coming from the aorta to suspect an associated pulmonary sequestration
Great read of tip’s with awesome guidance. Thank you for sharing this information.